Respen-A™: Homeopathic Treatment for Symptoms Associated With Autism
World's first successful treatment for the core symptoms of autism.
Set children free from the trap!
1 in 31 Children Is Diagnosed with Autism Spectrum Disorder (1 in 12.5 boys in the state of California). The risk for boys being diagnosed with ASD in the USA is 1 in 20.
ASD is a neuro-developmental condition usually manifested before the age of 3 years. It is four times more common in boys than girls.
Researcher Elaine DeLack (founder, MedDEV-OTC) has developed a scientific hypothesis for the cause of autism, and a homeopathic treatment that physicians and parents are using with success. To view a slide show outlining the scientific rationale for this hypothesis, click here. | List of Research References
OVERVIEW
Elaine DeLack, RN, CEO of MedDEV Inc., realized an urgent need for a treatment that successfully addresses all of the core symptoms of autism:
impaired Speech/Language
impaired Socialization
impaired Sensory/Cognitive Awareness
restrictive Health/Physical/Behavior
Please access the FAQ page for answers to most questions. If you have a new question, please ask us through our
FaceBook page.
Respen-A™ can be ordered directly through Elaine Delack without a prescription. To order, send an email to elaine.delack@gmail.com
STORY
Autism Spectrum Disorder (ASD) has reached epidemic proportions, increasing 647% in the last 20 years!
More children are diagnosed each year with ASD (1 in 36 children in the United States as of 2023, 1 in 22 children in the state of California) than with cancer, diabetes and AIDS combined!
These are our children- these children are our future!
Autism doesn't have to look this way...
Respen-A™, available since 2009, does not require a doctor's prescription. It can be ordered directly from the developer by sending an email to: elaine.delack@gmail.com
Of the 900+ families who have used Respen-A™ for their autistic children, most have only heard about it by word of mouth- there haven't been funds for costly advertising.
Thousands of hours of research have gone into the making of this product. Learned doctors and researchers, labs etc., have collaborated in producing this homeopathic treatment that is making a tangible difference in the lives of those suffering with ASD!
For such a time as this, when we can bring hope to many by making Respen-A™ more well-known, your support is greatly appreciated! Please tell others about Respen-A™! Don’t keep it a secret.
Together, we can change the face of autism!
Respen-A™ is Very Easy to Use
Easily Apply Dime-Sized Disc to Skin
Simply apply the topical disc to the skin in the morning and remove at bedtime. A Special occlusive and waterproof tape is included for this purpose.
Respen-A™ requires calcium supplementation. Note: The Respen-A™ 1.0 Disc can lower your intracellular calcium level, so it is very important that you take the recommended amount of calcium and magnesium every day! FAQ #7 for Respen-A™ thoroughly explains elemental calcium
Parents have reported noticeable improvements in core symptoms, some within ten minutes of applying the Respen-A™ disc on their child.
Endorsed by Doctors
More than 130 doctors have prescribed Respen-A™ to more than 900 patients with Autism Spectrum Disorder (ASD).
TV Worldwide Interview with Dr. Kurt Woeller about Respen-A
Respen-A™ Efficacy Proven in a Clinical Study
A recent study conducted by Dr. Anju Usman, MD, FAAFP, ABIHM, FMAPS, revealed a homeopathic treatment called Respen-A™ to have a significant effect in improving the core symptoms in autistic patients.
Dr. Usman, who specializes in Autism Spectrum Disorders (ASD), conducted a study of Respen-A™ in nine of her patients diagnosed with ASD. Dr. Usman utilized the Autism Treatment Evaluation Checklist (ATEC) , a validated tool developed by the Autism Research Institute to evaluate the effectiveness of a treatment to improve the core symptoms of ASD.
Researchers have used the ATEC to document improvement following intervention by comparing the baseline ATEC scores with the post-treatment ATEC scores. Lower scores indicate less severity of the symptoms. Eight of the nine patients showed significant improvements in Speech/Language/Communication, Sociability, Sensory/Cognitive Awareness and Health/Physical/Behavior while wearing the Respen-A™ topical disc.
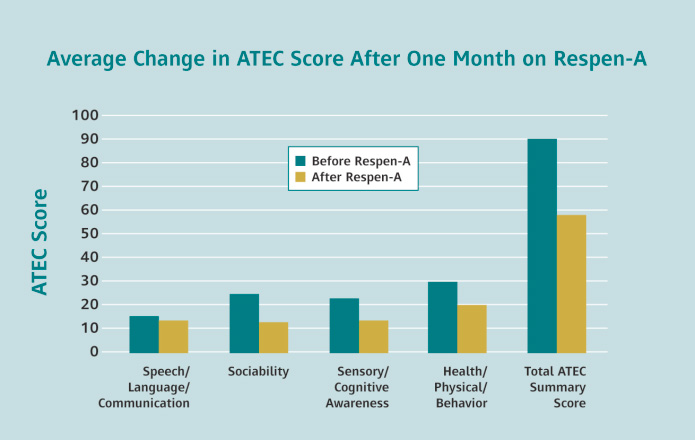
Despite the very small number of patients analyzed in the study, Respen-A™ showed a remarkably high effect with a p value of 0.001, meaning there was only a 1 in 1,000 possibility that the improvements seen with the Respen-A™ disc were simply by chance alone. A p value of 0.05 or less is considered significant, meaning there is only a 5 in 100 probability that the results were in error.
Dr. Usman stated, “These results were unexpected because of the small study size, the older age of the patients and the chronic nature of their issues that improved in just 3 months.” The statistician for the study concluded that Respen-A™ showed a statistically significant improvement in the core symptoms of autism, having a p value of 0.001, which in layman terms means that the likelihood of a false positive was 1 in 1,000.
Patients averaged 30 points of improvement in their symptoms of autism after one month of using Respen-A™. Many patients’ symptoms have resolved and are no longer considered on the Autism Spectrum after using the biomedical treatment, Respen-A™, less than one year.
Quantitative Electroencephalogram in autism
The Scientific Rationale Behind Respen-A™
Respen-A™ is not a cure for autism spectrum disorder (ASD), but treats the core symptoms:
impaired Speech/Language
impaired Socialization
impaired Sensory/Cognitive Awareness
restrictive Health/Physical/Behavior
Respen-A™ is a homeopathic dilution of reserpine, which is an old blood pressure medication. Reserpine has been shown to double the activity of the mitochondria enzyme, monoamine oxidase-A (MAO-A). Decreased MAO-A activity has been correlated with ASD.
Because reserpine is recognized as a homeopathic agent in the Homeopathic Pharmacopoeia of the United States (HPUS), Respen-A™ does not need to go through the typical FDA approval process for a drug.
 Mitochondria Enzyme, Monoamine Oxidase-A
Mitochondria Enzyme, Monoamine Oxidase-A
Meet Elaine, the Innovator of Respen-A™
Respen-A™ was developed by Elaine DeLack, RN, CEO and founder of MedDEV Inc. Elaine became involved in medical research out of a need to help herself after she was diagnosed with multiple sclerosis (MS), and the therapies medicine had to offer weren’t helping her.
After developing a successful treatment for Multiple Sclerosis that garnered national media attention, Elaine received a phone call from a mother who had seen her research in MS, and hoped she could help her son who suffered with severe autism [the child in the video below]. She told Elaine that she hadn't been able to hug her son for 7 years (when he was 3 years old) as he was so sensitive to touch that he couldn't even wear clothing. This compelled Elaine to begin years of biomedical research which resulted in a hypothesis that addresses the cause of autism and the rise in incidence of diagnosis, as well as a successful treatment to help free children from the trap of ASD.
Elaine will never forget the day the mother called her back, in tears, because her son, after just 4 days on Respen-A™, had reached up and hugged her. "I can't describe the joy I feel when I receive that kind of phone call- it makes everything worthwhile", says Elaine.
Meet Matt Whose Story Inspired the Innovation of Respen-A™
Notice how Respen-A™ helped his symptoms after just 3 days of use!
Touching Testimonial of a Child's Response to Respen-A™
Please read the following transcript from 7 year old Matthew, who had never spoken or written a word prior to using Respen-A™. Every week his teacher, Lisa, would try to get him to communicate using a computer, but she'd never had any response. It was questionable if Matthew even understood language. After 3 weeks on Respen-A™, he typed his first communication ever!

You Can Help Us Change the Face of Autism and
Free Children and Adults From the Trap!
With your support we can reach more children like Matthew, and set them free from the trap of autism. Please help us by pledging to support our endeavor, and please share the link to this grassroots campaign with your family and friends to help spread the word and bring hope to all families affected by autism!
FREE one month supply of Respen-A™ to New Users!
Elaine is now offering one month of Respen-A™, FREE to new users . This is a $50 savings!
We want every parent to experience the joy of recovering their child from autism, ADD/ADHD, anxiety, depression, bipolar disorder, and more, so tell your friends and people you know about this special offer!
You'll pay for shipping (shipped USPS Priority Mail in the U.S.). Order ships upon payment.
Please help us help others by telling your friends about this opportunity to try Respen-A™, FREE, and help us to change the face of autism! Tell them to send their request for this offer to Elaine at elaine.delack@gmail.com.

